|
Shell #2 has four higher energy orbitals, the 2s-orbital being lower in energy than the three 2p-orbitals. According to the Aufbau principle, the electrons of an atom occupy quantum levels or orbitals starting from the lowest energy level, and proceeding to the highest, with each orbital holding a maximum of two paired electrons (opposite spins).Įlectron shell #1 has the lowest energy and its s-orbital is the first to be filled. The truncated periodic table shown above provides the orbital electronic structure for the first eighteen elements (hydrogen through argon). Consequently, our understanding of organic chemistry must have, as a foundation, an appreciation of the electronic structure and properties of these elements. Other interactive periodic tables provide comprehensive data for each element, including nuclide properties, environmental and health factors, presentation in different languages and much more.įor comic relief you may wish to examine a periodic table linked to element references in comic books.įour elements, hydrogen, carbon, oxygen and nitrogen, are the major components of most organic compounds. There are, of course, over eighty other elements.Ī complete periodic table, having very useful interactive links has been created by Mark Winter. The periodic table shown here is severely truncated. This module introduces some basic facts and principles that are needed for a discussion of organic molecules.Įlectron Configurations in the Periodic Table 1A 
The study of organic chemistry must at some point extend to the molecular level, for the physical and chemical properties of a substance are ultimately explained in terms of the structure and bonding of molecules. Show Electronic configuration of Neon.Electron Configurations & The Periodic Table no electron is available for bondingĢ) It is mainly use to make switching gears and diving equipmentģ) Neon is important component in cryogenic refrigeratorĥ) It is use in lung diffusion test as a tracker gas Neon show 0 valency Because it’s octate is completeĤ) Atomic radius : Atomic radius of neon is 38 pmĥ) Reactivity : It is non reactive element because it’s outer most shell is full. Atomic mass is 21Įlectronic configuration of neon is 1S 2 2S 2 2P 6Įlectronic configuration in concert of shell is 2,8Ģ electron present in K shell and 8 electron present in M shell. Atomic mass is 19Ģ1Ne – it is present 0.27%. 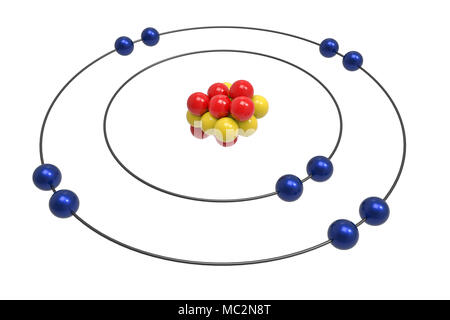
Origin of name : The name is derived from Greek word’ neos’ Meaning of neos is newġ) Atomic symbol : Atomic symbol of neon is NeĢ) Atomic number Atomic number of neon is 10ģ) Atomic weight : Atomic weight of neon is 20.17ġ0 proton and 10 neutros present in the neon atomĤ) Position : It is placed in 2 row ( period ) and 18 column( group) It is known as Nobel gas.ģ stable isotopes and remaining are radioactive isotope 20Ne – it is present 90%. neon and halogen is separatedĭiscovery : It is discovered in 1898 by William Ramsay and Morris Travers. It is derived from using fractional distillation. It is 5 th mostly found element in earth surface. When it is burn in tube it glow as a red. 
It is known as zero gas It is non toxic gas. Neon is an inert gas present in monoatomic from. Atomic Mass, Number, Physical, Chemical properties, Electronic configuration, Valency, Chemical reaction, Uses Neon – Learn all details regarding Neon in Periodic Table i.e.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
